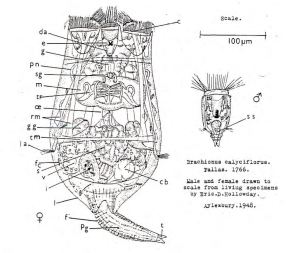
On Saturday 30 January, Roger Hawkins and I are running a workshop for the BENHS on using keys for insect identification. We’ll mostly be working with published hard-copy keys, but we’ll include some online keys as well. Below are some examples for various invertebrate groups.
On Saturday 30 January, Roger Hawkins and I are running a workshop for the BENHS on using keys for insect identification. We’ll mostly be working with published hard-copy keys, but we’ll include some online keys as well. Below are some examples for various invertebrate groups.
I must admit that I don’t yet make much use of online keys. This is partly because I’m fortunate in having a good range of printed keys available to me, and I’m sure it’s also partly a case of ‘sticking with what I know’ rather than spending time learning the online ones (all keys require time spent on them to get familiar with their particular approach). However, I think there are still some problems with online keys, from the practical one of having to have a computer within easy view of your specimen or microscope, to more intrinsic issues about the ease of flicking through a book to compare different sections, as opposed to having to switch between different windows on a computer.
However, online keys are likely to become more widespread and will no doubt get better as time goes on, just as printed ones have (and continue to do so), and no doubt the two will be seen as complementary rather than an either/or choice.
See below for some online keys to try out. For a longer list (including plants and other groups as well as invertebrates) see my bookmarks on delicious, and let me know if there are any other good keys out there, or any thoughts you have on using online keys. A post on downloadable keys will follow.
Various groups
The most comprehensive set of online keys that I am aware of is the DELTA Intkey system. This requires you to download some software onto your computer, after which you can either download various individual keys, or run them from the web.
At the moment there is quite a wide range of keys available within this system, for various taxonomic groups (not just invertebrates) and different parts of the world, but for UK insect purposes they include:
Orders of insects, families of Coleoptera, families of Diptera, genera of Ephemeroptera, families of Hemiptera, families of Hymenoptera, families of Lepidoptera, genera of butterflies, genera of Geometridae, genera of Noctuidae, species of Phyllonorycter, species of Odonata, genera of Orthoptera, families of Trichoptera.
These are multi-access keys so that you can answer the set of questions in any sequence, and need only answer the ones you’re confident of – the system will endeavour to give you a best match of one or more names for your specimen. I’ve made most use of the family keys for Coleoptera and Diptera, but I have to admit they’ve not proved as helpful as I hoped, and I still tend to return to paper-based single-access (dichotomous) keys for backup. But it is always good to have additional keys available for comparison, and no doubt if I used them more I’d get more used to their idiosyncrasies and perhaps find them more helpful.
There are some draft online keys available on iSpot (part of OPAL), including a simple ‘key to minibeasts’ – this part of iSpot is still under development, and there’ll be more to come.
Coleoptera
The Watford Coleoptera Group (click on “ID aids”) are making a range of identification aids available, some in the form of keys, some as notes on particular species or groups of species.
Hymenoptera
[added on 10 February 2010]
The Natural History Museum provides a very useful key to bumblebees.
Diptera
These online keys to various families of Diptera are managed by Paul Beuk, who also runs the excellent Diptera.info. You may need to register on Diptera.info to get full access to the keys. Some of these keys are online versions of existing printed keys, others are new (e.g. includes the best key I’m aware of for genus Sylvicola in family Anisopodidae).
Not a key, but some very helpful support for keys is provided by the Anatomical Atlas of Flies, from CSIRO in Australia. This is a truly excellent web implementation of a morphology diagram and glossary of names for parts of flies, using detailed close-ups of real insect specimens. But it needs a good broadband connection to work at any speed!
Mark van Veen’s Faunist is a Dutch site with keys to various families of Diptera, plus Odonata, Orthoptera and sea-shells. The latter three are in Dutch only, but most of the Diptera keys are in English. They are well-illustrated and easy to use, and I think cover most of the British species in the families included. Plenty of information about the species is given (but remember that this refers to the fauna in Holland, which will include additional species and different habitats/behaviours compared to the UK).
Some of these keys to robber-flies (Asilidae) by Fritz Geller-Grimm are applicable to the UK (others cover various parts of the world).
For those flies with larvae that produce leaf-mines (largely Agromyzidae, plus a few Anthomyiidae etc.) there are keys based on the foodplant on the excellent UK Fly Mines site.
Lepidoptera
Similarly, for leaf-mines of Lepidoptera try Barry Dickerson’s online key, on the British Leafminers website. This is largely based on volumes 1 and 2 of “The Moths and Butterflies of Great Britain and Ireland”, but with some additions and revisions. A very useful resource (but be aware of all the non-lepidopterous insects that also make leaf-mines, most of which are listed, but not keyed, elsewhere on the site).
Butterfly Conservation/Moths Count provide a simple key to day-flying moths.
Odonata
There is a good identification key to Irish dragonflies and damselflies, but it covers males only, and not all UK species are included.
Mollusca
The Conchological Society provide some online keys and other identification notes for various groups of snails and slugs.
Arachnida
This German website has a key to European spider families.